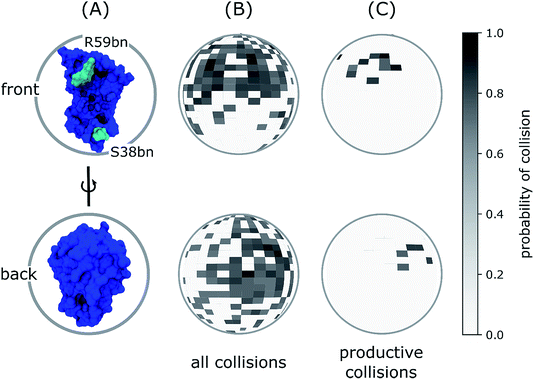 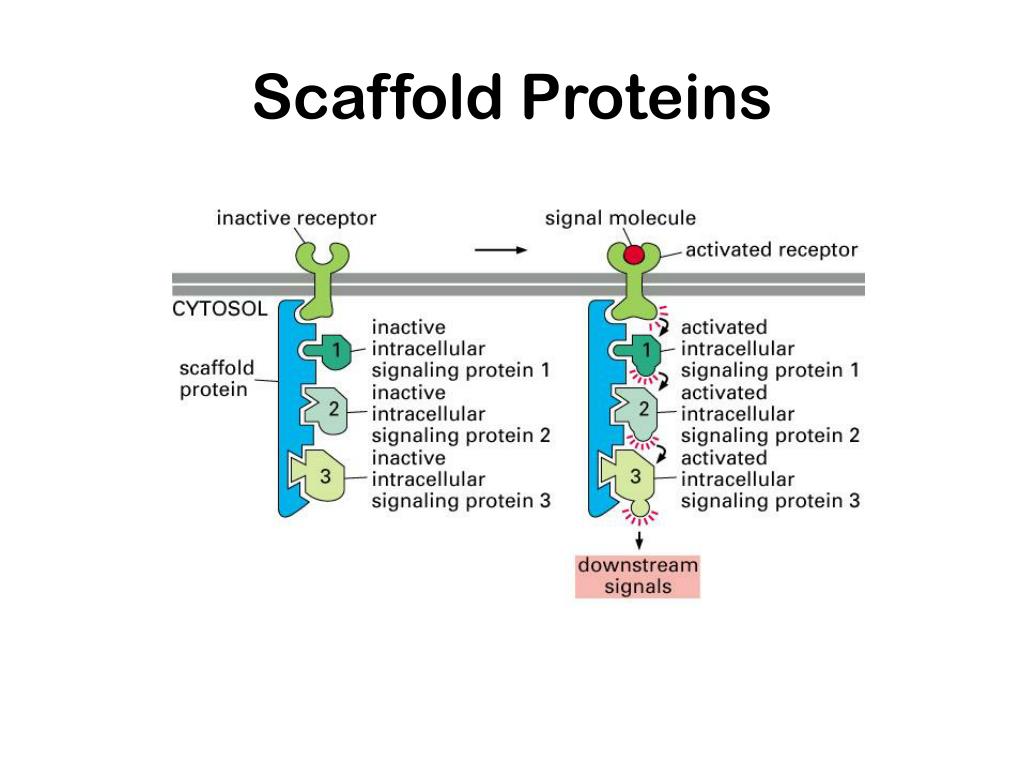
A common example of how scaffolds enhance specificity is a scaffold that binds a protein kinase and its substrate, thereby ensuring specific kinase phosphorylation. This assembly may be able to enhance signaling specificity by preventing unnecessary interactions between signaling proteins, and enhance signaling efficiency by increasing the proximity and effective concentration of components in the scaffold complex. Scaffolds assemble signaling components of a cascade into complexes. This particular function is considered a scaffold's most basic function. Scaffold proteins act in at least four ways: tethering signaling components, localizing these components to specific areas of the cell, regulating signal transduction by coordinating positive and negative feedback signals, and insulating correct signaling proteins from competing proteins. Three distinct domains of Ste5 were shown to associate with the protein kinases Ste11, Ste7, and Fus3 to form a multikinase complex. The first signaling scaffold protein discovered was the Ste5 protein from the yeast Saccharomyces cerevisiae. In such pathways, they regulate signal transduction and help localize pathway components (organized in complexes) to specific areas of the cell such as the plasma membrane, the cytoplasm, the nucleus, the Golgi, endosomes, and the mitochondria. Although scaffolds are not strictly defined in function, they are known to interact and/or bind with multiple members of a signalling pathway, tethering them into complexes. Even more specifically, the invention relates to a functional fusion of a toxin and a scaffold protein wherein the folded scaffold protein interrupts the topology of the toxin by insertion in an exposed β-turn of a β-strand-containing domain of said toxin to form a rigid fusion protein that retains its high affinity target binding capacity.In biology, scaffold proteins are crucial regulators of many key signalling pathways. More specifically, the present invention relates to novel fusion proteins, their uses and methods in three-dimensional structural analysis of macromolecules, such as X-ray crystallography and high-resolution Cryo-EM, and their use in structure-based drug design and screening, and as pharmacological tools. Even more specifically, the invention relates to a functional fusion of a toxin and a scaffold protein wherein the folded scaffold protein interrupts the topology of the toxin by insertion in an exposed β-turn of a β-strand-containing domain of said toxin to form a rigid fusion protein that retains its high affinity target binding capacity.ĪB - The present invention relates to the field of structural biology and drug discovery. N2 - The present invention relates to the field of structural biology and drug discovery. T1 - Fusion protein with a toxin and scaffold protein
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
